The FDA Bait-and-Switch on Pfizer Covid-19 Jab

By The Sharp Edge
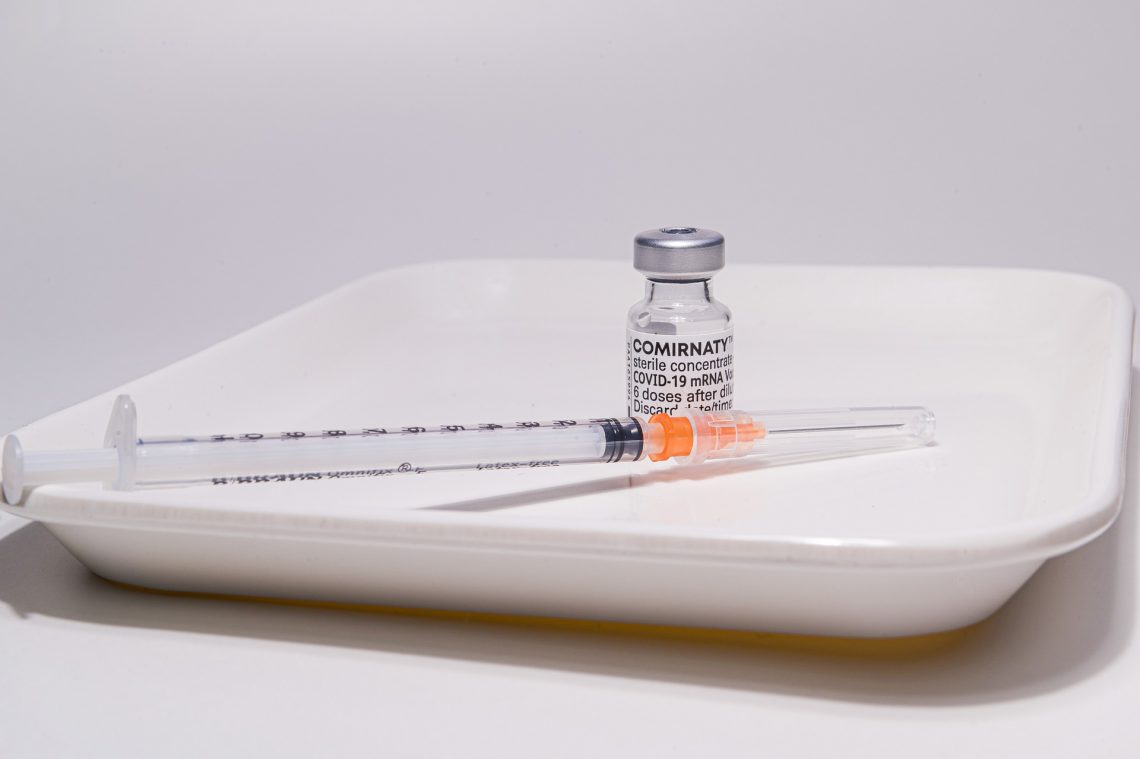
On August 23, 2021, the FDA announced their approval of the first Covid-19 jab. “The vaccine has been known as the Pfizer-BioNTech COVID-19 Vaccine, and will now be marketed as Comirnaty (koe-mir’-na-tee), for the prevention of COVID-19 disease in individuals 16 years of age and older. The vaccine also continues to be available under emergency use authorization (EUA), including for individuals 12 through 15 years of age and for the administration of a third dose in certain immunocompromised individuals,” the news release stated.
The legacy media quickly picked up their talking points and ran with them. Soon after, mandates swept across the public, private and academic sectors. “Let me say this loudly and clearly,” Biden announced, “If you’re one of the millions of Americans who said that they will not get the shot…until it has full and final approval of the FDA, it has now happened. The moment you’ve been waiting for is here. It’s time for you to go get your vaccination and get it today.” Understandably so, the public has been led to believe that the jab currently on the market has full FDA approval – which is nothing short of a bait-and-switch.
While the FDA did offer BLA approval for BioNTech’s Covid-19 jab labeled “COMIRNATY,” (which is currently in limited supply in the United States) the Food and Drug Administration also provided an extension of the EUA for the Pfizer BioNTech Covid-19 jab (of which there is still a significant amount in stock.) The FDA also authorized “use of COMIRNATY (COVID-19 Vaccine, mRNA) under this EUA for certain uses that are not included in the approved BLA.” Furthermore, Pfizer’s fact sheet states that “The FDA-approved COMIRNATY (COVID-19 Vaccine, mRNA) and the EUA-authorized Pfizer-BioNTech COVID-19 Vaccine have the same formulation and can be used interchangeably to provide the COVID-19 vaccination series.”
There are now two seemingly identical products that are “legally distinct” – one jab which is currently available under EUA, and another FDA approved jab which is not widely available to the American public. It is unclear when, if ever, the FDA licensed version of Comirnaty will be fully in stock in the United States. It could be weeks, months, or even later, although there have been some reports that jabs labeled “Comirnaty” are already in circulation in Europe and elsewhere. As long as an EUA for the same product exists, it seems that Pfizer has little incentive to quickly jump through the necessary stringent legal hoops required for their Comirnaty jabs with the FDA stamp of approval clearly marked on the labeling. Disclosures on any biological product deviations, adverse events, or content of labeling (as required in the FDA approval letter) may be reasons for delay. According to the CDC, as of August 18, 2021, there have already been 6,968 deaths after receiving a Covid jab, reported through their VAERS system.
As for the currently available jabs offered under the extended EUA issued to Pfizer, “All descriptive printed matter, advertising, and promotional material relating to the use of the Pfizer-BioNTech COVID‑19 Vaccine” (presumably whether they are labeled “Comirnaty” or not) must “clearly and conspicuously state” that:
• This product has not been approved or licensed by FDA, but has been authorized for emergency use by FDA, under an EUA to prevent Coronavirus Disease 2019 (COVID-19) for use in individuals 12 years of age and older; and
• The emergency use of this product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of the medical product under Section 564(b)(1) of the FD&C Act unless the declaration is terminated or authorization revoked sooner.
Why would the FDA make an “approval” so unclear? If this FDA approval was an attempt to establish public trust, they have failed miserably. Now, with help from the propaganda arm of Big Government and Big Pharma, their message has broadcast across the nation: “Pfizer’s Covid vaccine is FDA approved. Let’s start mandating it.” The government and pharmaceutical companies are well aware that most people are not trained in deciphering double-talk legal jargon and likely won’t think of checking the actual labels. Like practically everything Covid related, the government has managed to make this whole “FDA approval” process as clear as mud. Now millions of Americans who are facing pressure from mandates by schools and employers find themselves sifting through murky legalese while asking, “Is the jab they are trying to force on me FDA approved or not?”
Since the announcement of this so-called approval, well-informed critics such as Robert F. Kennedy, Jr., have been scouring the fine print and raising glaring concerns, which have been purposefully overlooked by the mainstream media. In an article co-authored by RFK, Jr. entitled ‘2 Things Mainstream Media Didn’t Tell You About FDA’s Approval of Pfizer Vaccine,’ it explains, “the FDA pointed out that the licensed Pfizer Comirnaty vaccine and the existing, EUA Pfizer vaccine are ‘legally distinct,’ but proclaims that their differences do not ‘impact safety or effectiveness.’ There is a huge real-world difference between products approved under EUA compared with those the FDA has fully licensed. EUA products are experimental under U.S. law. Both the Nuremberg Code and federal regulations provide that no one can force a human being to participate in this experiment. Under 21 U.S. Code Sec.360bbb-3(e)(1)(A)(ii)(III), ‘authorization for medical products for use in emergencies,’ it is unlawful to deny someone a job or an education because they refuse to be an experimental subject. Instead, potential recipients have an absolute right to refuse EUA vaccines.” The simultaneous FDA license of Comirnaty and EUA extension of virtually the same jab in all but legality seemingly attempts to blur the lines of this fundamental issue that impacts millions of Americans.
Under Emergency Use Authorization the Pfizer jab is shielded from liability, while key questions have been raised regarding their liability under the FDA approved version of the jab. It appears that Pfizer will be able to exploit yet another legal loophole due to the Public Readiness and Emergency Preparedness Act (PREP Act). In March of 2020, the PREP Act was enacted to “provide liability immunity for activities related to medical countermeasures against COVID-19”. The Act implies that it provides immunity from liability for all vaccines regardless of their FDA approval status. Importantly, this declaration of immunity excludes cases of “willful misconduct.” The PREP Act established that a compensation program called the Countermeasures Injury Compensation Program (CICP) will be used for Covid jab related injury claims. According to their latest data as of August 2, 2021, CICP has received 686 injury or death claims from Covid-19 vaccines and has yet to award any compensation for those claims. While the public may have limited ability to seek compensation for injury through the CICP program, it looks like Pfizer will be protected from liability for both their EUA and FDA approved jabs. How exactly are these Herculean efforts to shield the pharmaceutical giant from liability supposed to instill confidence in the public?
As with every aspect of this entire Covid agenda, misleading the public has been the name of the game. The latest FDA bait-and-switch is no different. Their scheme is clearly designed to provoke businesses and schools into the enforcement of Covid jab mandates, while falsely leading the American public to believe the mandates are legal. They may have succeeded in fooling some thousands of unsuspecting people into succumbing to the pressures imposed by mandates due to this legal trickery, but if the entire point of this exercise was to instill confidence in an already broken and corrupt system – undoubtedly millions remain justifiably skeptical.

Subscribe to Corey’s Digs so you don’t miss a Dig!










18 Comments
greg
thanks again corey. more going on than meets the eye as usual. it was in my mind that the timing of the fda approval made it appear as though the sudden fda approval was nothing but political and thereby melted away any last vestiges of credibility that the fda and upper echelon medicine may have had left. but i now understand that it was even more than that. fda is still a politicial shill, but on a broader scope than i had imagined. oh we do live in interesting times; don’t we?
Jeff Buckshorn
Corey~ I wish you would listen to a Dr. David Martin (I saw him on forbidden knowledge.tv) and do a deep dive on Dr. FAUCI? In an hour and 1/2 this Dr. tells you about the patents,etc.,and the CONTROL that SOB has over everyone and how he and others are KILLING us all! MUST READ OR LISTEN!
David Robertson
Even if such Jabs were FDA approved, or in my case approved by Health Canada, I would still not take them. My trust in any government or government agency is zero and has been for many years. I have had no cold or ‘flu for decades, indeed I cannot remember when I last had one.
I have used natural supplements to maintain my health for the past 16 years and am reasonably healthy while having Type 2 diabetes for the past 21 years. I used to use Metformin to control my blood sugar levels but switched successfully to natural remedies some years ago.
I am very leery of the official health system since losing my dear wife to the NHS in Scotland in 2015. They kidnapped and killed her with drugs designed for that purpose over my protests and ignored my request to release her into my care. I have since seen a video on youtube by Dr. Amanda Vollmer describing a similar situation recently with one of her patients whose relatives had to fight to rescue him from these medical murderers who were in the process of “putting him down”. They were successful where I failed.
Wendy Litchfield
Thank you for putting this out there for everyone. I read the 14 page doc last week confirming this. I also have confirmation from an email from FDA that all the covid tests are EUA as well.
Paula
So I’m being forced into getting the job or being tested every week I work for school system and I don’t agree with any of this what do I do?
SoulSearcher
1 thing I think you missed is that for an emergecy use product to be approved, there has to be no other approved drug out there. In others words there is no other option. So if there is an approved one, then they have to take all the emergency use ones off the market. There is no debate on that, because its the law. And that’s not happening. The FDA is violating the law in one way or another, by having emergency use jabs on the market, while there’s an approved jab on the market at the same time. Also this is being challenged by Robet Barnes. (lawyer) He said he was going to file the lawsuit this last Monday. Either the FDA approved one and left the others on the market
which violated the law, or they lied and didn’t approve anything. You can’t legally have it both ways.
Nancy Plummer
You explained this bs FDA action very well. Thank you.
Mary
FDA Approval of COVID-19 Vaccine Is a Scam – Robert Barnes, Sep 1, 2021
https://sageofquayblog.blogspot.com/2021/09/robert-barnes-fda-approval-of-covid-19.html
Attic7
Most people –including scientists, academics, doctors, and other formally educated folks– are rather profoundly perplexed and befuddled by the various absurdities of the Covid crisis (e.g., the back-and-forth shifting of scientific decrees, the censorship of valid data, the presence of toxic graphene oxide nanoparticles in Covid vaccines, etc.). They cannot really make sense out of it all. At best, they can DESCRIBE the absurdities but they cannot explain why it is happening.
Why is that? It is because they lack pieces of vital knowledge, whose lack hinders and disables accurate coherent full understanding (therefore, it impairs the proper decision-making process and the potential for profound constructive action).
Those “missing” pieces of knowledge right in front of our noses are described in a comprehensive article called “The 2 Married Pink Elephants In The Historical Room –The Holocaustal Covid-19 Coronavirus Madness: A Sociological Perspective & Historical Assessment Of The Covid “Phenomenon”” by Rolf Hefti at https://www.rolf-hefti.com/covid-19-coronavirus.html
Without a proper understanding, and full acknowledgment, of the true problem and reality, no real constructive change is possible.
GoAlive
Here you find the Dossier of Mr. Martin
https://www.corona-ausschuss.de/wp-content/uploads/2021/07/FauciDossierWordFileText.pdf
GoAlive
“The licensed vaccine has the same formulation as the EEA-authorized vaccine and the products can be used interchangeably to provide the vaccination series without presenting any safety or effectiveness concerns. The products are legally distinct with certain differences that do not impact safety or effectiveness.”
This is a great sequence, as only great lawyers are capable of formulating!
What does “same formulation” mean and, above all, what does “certain differences” mean?
Laymen assume that then the identical is also in it – but it doesn’t say that!
I have looked into the components of the socalled “mRNA vaccines” and can say that on the one hand it is about an NIH patented mRNA for the spike protein, which Moderna and BioNTech use, and on the other hand about nanolipids (LNP) which are different and are produced by Acuitas and SINOPEG:
https://de.sinopeg.com/sinopeg-covid-19-vaccine-excipients-supply_n33
But details on how Moderna arrived at its optimal formulation in the first place are scant. The company did not grant an interview to talk about its nanoparticle development, and neither did Pfizer or BioNTech. For its COVID-19 vaccine, Moderna ultimately used an ionizable lipid that it calls SM-102, which it first described in a 2018 study on alternatives to MC3. Pfizer and BioNTech licensed an ionizable lipid called ALC-0315 from Acuitas
https://cen.acs.org/pharmaceuticals/drug-delivery/Without-lipid-shells-mRNA-vaccines/99/i8
Medical experts and molecular biologists say that the dangers for the vaccinated are less from the mRNA than from these LNPs, which are considered “excipients.” Suspicious medical experts also say that the whole vaccination campaign is about testing these LNP, which can also be used for body ID and programming, and not about the mRNA, which is actually incidental to it.
Therefore, I strongly assume that the “certain differences” refer to exactly these LNP, which have the task to shape a transported object, which here happens to be this mRNA (“same”), in such a way that it is tolerated by the body. However, corresponding experiments on animals were unfortunately always unsuccessful.
Gems Gram
No ‘vaccine’ has EVER been proven effective or safe! If it comes from Big Pharma, JUST SAY NO!
Susan Volwiler
Interesting how they co-mingled the word “comity,” meaning social harmony, with RNA.
Use critical thinking people
96% of doctors are vaccinated against covid 19. 96% people. Read that again. 96%.
Doc Casper
US has over one million physicians and over 800,00 in daily practice. Only 301 physicians responded to the 5 day AMA survey. Representatve sample?
Rusty
David Robertson, can you tell me what natural supplements you replaced metformin with? I am a type 2 diabetic as well. Thank you.
Arnie Willis
Corey have you done any digging into HOW BAD IS MY BATCH and it’s link to where they say the deadly batches with lot numbers are going. They say the deaths are highest in republican areas. The there HOWBAD.COM. Thanks love you guys and your work.
Corey Lynn
I published this a couple weeks ago, which includes what you are talking about. I haven’t dug into the logistics of them being in Republican areas, but I have seen some charts on that after I published this article: https://www.coreysdigs.com/health-science/pfizer-fda-corruption-lethal-batches-and-autopsies-reveal-covid-19-jab-genocide/